Glucagon-like peptide-1 (GLP-1) receptor agonists (RAs) are an emerging class of glucose-lowering drugs that are increasingly used in the treatment of type 2 diabetes (T2D) and obesity. Cardiovascular outcome trials in patients with T2D have demonstrated the efficacy of GLP-1 RAs in reducing major adverse cardiovascular events, including cardiovascular death, MI and stroke, regardless of glycaemic control.1-8
Recent European Society of Cardiology guidelines for the management of cardiovascular disease (CVD) in patients with diabetes state that GLP-1 RAs (lixisenatide, liraglutide, semaglutide, exenatide extended release [ER], dulaglutide, efpeglenatide) should be considered for glucose-lowering treatment in patients with T2D at risk of or with heart failure (HF; class IIa A recommendation) already taking sodium–glucose cotransporter 2 inhibitors, without differentiating between HF phenotypes.9 The consensus statement from the American Association of Clinical Endocrinologists supports the use of GLP-1 RAs as first-line therapy in managing hyperglycaemia in T2D patients with established (or at high risk of) atherosclerotic CVD, as well as in those with chronic kidney disease or a history of stroke or transient ischaemic attack, while recommending the administration of sodium–glucose cotransporter 2 inhibitors to HF patients.10 The American College of Cardiology/American Heart Association primary prevention of cardiovascular disease guideline supports the use of GLP-1 RAs in patients with T2D and high atherosclerotic CVD risk, but does not mention existing HF.11
Current evidence related to the effects of GLP-1 RAs on HF outcomes remains limited. GLP-1 RA cardiovascular outcome trials in patients with T2D reported a neutral impact of the drugs on HF hospitalisation. Of note, the prevalence of HF varied across these trials from 9% to 24%, and HF events were considered a secondary endpoint.1–8 Most of these studies do not report HF diagnostic criteria or mention HF therapy, and only one trial reported on left ventricular ejection fraction (LVEF) at baseline.8 Moreover, one should consider the fact that different GLP-1 RAs were used across the studies, with different chemical structures, durations of action and weight-lowering effects, which may impact their efficacy.
An updated meta-analysis of nine randomised controlled trials (RCTs), including 8,920 patients with HF and T2D, reported a 13% reduction in major adverse cardiovascular events in the GLP-1 RA compared with placebo arm.12 In contrast, no benefit of GLP-1 RAs was observed in terms of all-cause death, HF hospitalisation or cardiovascular death.12 Of note, that meta-analysis did not differentiate between HF phenotypes.
In the recent STEP HFpEF DM trial, which studied the efficacy of semaglutide among patients with obesity-related HF with preserved ejection fraction (HFpEF) and T2D, a reduction in the time to first HF events was demonstrated in the intervention arm, but this was a secondary endpoint.13 No RCTs have primarily tested the effects of GLP-1 RAs on HF hard outcomes and/or mortality in patients with HFpEF or HF with reduced ejection fraction (HFrEF).
Generally, surrogate endpoints are expected to predict clinical benefits. In recent years, data have been accumulated suggesting effects of GLP-1 RAs on surrogate HF outcomes in HF patients across LVEF, including cardiac structure and function, exercise capacity and quality of life.13
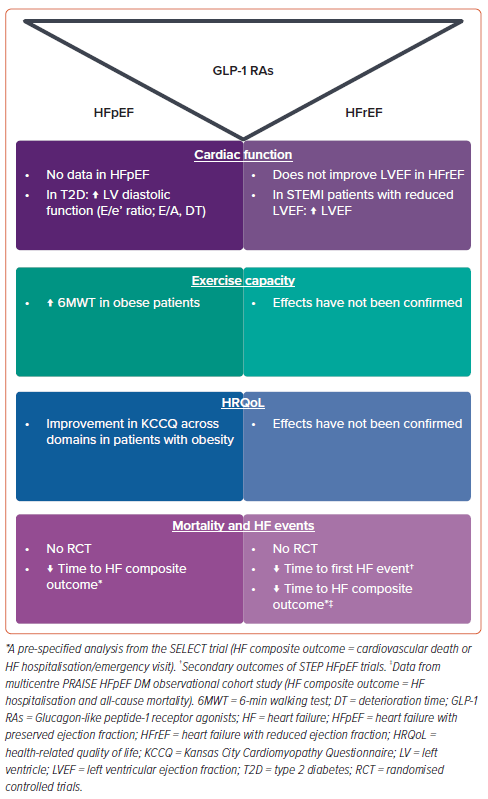
In this review, we summarise the evidence related to the effects of GLP-1 RAs on these outcomes (Figure 1), which may shed light on the potential of these agents to be used in the clinical care of HF patients and investigated in large clinical trials to evaluate clinical outcomes.
Potential Mechanism Underlying the Effects of GLP-1 RAs in Heart Failure
GLP-1 RAs bind to GLP-1 receptors expressed in various tissues, including pancreatic beta cells, the kidneys, heart, brain and gastric mucosa, among other organs. After binding to GLP-1 receptors in pancreatic beta cells, GLP-1 RAs exert their glucose-lowering effect by stimulating glucose-dependent insulin release. Antihyperglycemic effects aside, GLP-1 RAs reduce gastric emptying and suppress appetite centres in the hypothalamus, resulting in weight loss. Systemic effects of GLP-1 RAs, mediated via GLP-1 receptors in other organs and systems, include enhanced endothelial function and myocardial metabolism, natriuresis and anti-inflammatory and blood pressure-lowering effects.14–17
All these effects of GLP-1 RAs are crucial for the pathophysiology of HF, with weight loss in obese HF patients being of utmost importance. Obesity is highly prevalent in HF; in HFpEF, the prevalence of obesity reaches up to 80%.18 Obesity is an important contributor to the pathophysiology of both diabetes and HF, particularly HFpEF.19 Adipose tissue is a complex endocrine organ that has multiple endocrine and paracrine effects on the heart.20 Moreover, the high amount of visceral fat and epicardial adipose tissue (EAT), as well as associated plasma volume expansion, in obese individuals results in significant haemodynamic impairment at rest and during exercise, exerting local effects on the heart.21 Remarkably, even in HF-free patients with morbid obesity, cardiac abnormalities can be seen that are consistent with left ventricular (LV) remodelling and dysfunction, including greater LV mass, LV diastolic dysfunction and elevated LV filling pressure.22,23
Obese HFpEF patients have greater LV hypertrophy and pulmonary capillary wedge pressure, increased LV filling pressures and severe right ventricular dysfunction compared with non-obese HFpEF patients.24,25 Increased EAT also results in a greater pericardial restraint, which affects haemodynamics.26 Overall, the cardiovascular effects of GLP-1 RAs in HF can be realised through multiple pathways, including reducing visceral fat and EAT in obese HF patients; abolishing obesity-related haemodynamic impairment, pathological LV remodelling and myocardial inflammation; and systemic and glucose-lowering effects.27
Effects of GLP-1 RAs on Cardiac Structure and Function
GLP-1 RAs have garnered significant interest due to their potential benefits beyond glycaemic control, including their effects on cardiac function.28 A landmark pilot study that investigated the efficacy and safety of 72-h GLP-1 RA infusion in patients with acute MI and impaired LVEF (<40%) revealed a notable improvement in LV function, as assessed by echocardiography, compared with control subjects with similar disease characteristics that was independent of diabetes.29 Furthermore, in a randomised placebo-controlled trial involving patients with ST-segment elevation MI (STEMI) who underwent balloon angioplasty and stent placement, with daily injections of exenatide or placebo for 3 days, revealed exenatide was associated with a smaller infarct size on cardiac MRI conducted 38 days after reperfusion.30 Echocardiographic assessment at 6 months demonstrated persistent improvements in diastolic function and global longitudinal strain among patients who received exenatide.30 The reduction in infarct size was confirmed in a larger study of 172 STEMI patients randomised to receive either intravenous exenatide or placebo.31,32 However, in that study, no significant difference in changes in LVEF were observed between the groups.31,32 In contrast, the administration of liraglutide to STEMI patients was associated with a small but significant improvement in LVEF assessed at 3 months in both non-diabetic and diabetic subjects.33
Although GLP-1 RAs have shown promise in enhancing postischemic LV systolic function in preclinical and clinical settings, studies investigating the effects of GLP-1 RAs on LV diastolic function in patients with T2D reported conflicting findings.31,34–37 Several small studies reported that liraglutide significantly improved LV diastolic function in T2D patients compared with either placebo or other glucose-lowering agents, which was also associated with improvements in endothelial function and antioxidant and anti-inflammatory activity.36,37 In contrast, the administration of exenatide was not associated with changes in LV diastolic function.5 These apparent discrepancies could be explained, in part, by the drug-specific effects of GLP-1 RAs on LV diastolic function, as well as differences in the baseline characteristics of patients, namely less prominent LV diastolic dysfunction in patients in the exenatide cohort compared with patients in the liraglutide studies.5,36,37
However, a meta-analysis of 10 placebo-controlled RCTs, including 732 individuals with T2D, found that liraglutide therapy did not influence echocardiographic parameters of diastolic function compared with placebo, including the ratio of early diastolic filling velocity (E) to mitral annular early diastolic velocity (e′; weighted mean difference [WMD] −0.763; 95% CI [−2.157, 0.630]; p=0.283), change in e′ (WMD −0.069; 95% CI [−0.481, 0.343]; p=0.742) and change in E/e′ (WMD −0.683; 95% CI [−1.663, 0.298]; p=0.172).35 LVEF also remained unchanged with liraglutide therapy compared with placebo (WMD −0.651; 95% CI [−1.649, 0.348]; p=0.202).35
A further meta-analysis of 22 RCTs that included a considerably larger cohort (n=61,412) of T2D patients with or without cardiovascular disease and patients with cardiovascular disease alone revealed that treatment with GLP-1 RAs led to improvements in diastolic function (E-wave; standardised mean difference −0.40; 95% CI [−0.60, −0.20]; p<0.001), early diastolic to late diastolic velocities ratio (WMD −0.10; 95% CI [−0.18, −0.02]; p=0.01), E/e′ ratio (WMD −0.97; 95% CI [−1.54, −0.41; p<0.001) and E-wave deceleration time (WMD −9.96 ms; 95% CI [−18.52, −1.41 ms]; p=0.02), although LVEF was not affected.38
The effects of GLP-1 RAs on cardiac structure and function were also investigated in HF patients. An early small study of 12 HF patients with New York Heart Association (NYHA) Classes III–IV showed a significant increase in LVEF following 5 weeks of continuous subcutaneous infusion of GLP-1 RAs.39 However, larger studies have not demonstrated a significant effect of GLP-1 RAs on LV function, including albiglutide, which was administered over a long period (>12 weeks) to non-diabetic, overweight or obese individuals with HF (NYHA Class II–III) and LVEF below 40%.40 In the FIGHT trial, long-term administration of liraglutide to HF and HFrEF patients with or without diabetes did not significantly improve LVEF after 24 weeks of treatment.41 However, there was an increase in heart rate and more serious cardiac events, such as arrhythmias and acute coronary syndrome, in patients treated with liraglutide.41 In a meta-analysis of nine RCTs involving 8,920 patients with HF and coexisting T2D, GLP-1 RAs did not improve LVEF, LV end-diastolic volume or LV end-systolic volume.12
Although larger-scale studies are yet to explore the effects of GLP-1 RAs on cardiac structure and function, available evidence suggests that GLP-1 RAs may improve systolic and diastolic function in individuals with T2D who are at high risk of CVD and reduce infarct size after acute MI.33–37 In individuals with HFrEF, GLP-1 RAs do not affect LV systolic function, whereas the effects of GLP-1 RAs in individuals with HFpEF have not yet been investigated.
Effects of GLP-1 RAs on Exercise Capacity
It may be assumed that GLP-1 RAs could potentially increase exercise capacity primarily by promoting weight loss. Other mechanisms underlying the effects of GLP-1 RAs on exercise capacity include improvements in myocardial energetics, enhanced endothelial function, reductions in systemic inflammation and oxidative stress and modulation of skeletal muscle metabolism.14–17
Overall, clinical evidence regarding the effects of GLP-1 RAs on exercise capacity is limited and inconsistent, and varies across the spectrum of HF.42 The effects of GLP-1 RAs in HFrEF have been studied in several small trials. In FIGHT, there was no significant effect of liraglutide on 6-min walk test (6MWT) distances compared with placebo.40 In the LIVE trial, which evaluated the effects of liraglutide on LV function in stable chronic HF patients with and without diabetes, at the end of treatment patients from the liraglutide group were able to walk 28 ± 65 m longer during the 6MWT, compared with 3 ± 89 m for patients in the placebo group, with a mean difference of 24 m.43 However, more patients in the liraglutide than placebo group experienced serious cardiac adverse events, including significant arrhythmia.43 A similar trend towards a higher risk of unfavourable outcomes was observed in the post hoc analysis of the FIGHT trial.40
Another small trial compared the effects of 12 weeks’ treatment with albiglutide (n=27) to placebo (n=30) on cardiac function, cardiac metabolism and exercise capacity in HFrEF.43 Albiglutide did not improve myocardial glucose use or myocardial oxygen consumption, cardiac efficiency or the 6MWT distance. Surprisingly, a slight improvement in change of peak oxygen consumption (peak VO2) was observed in the albiglutide group compared with placebo (mean 0.9 ± 0.5 ml/kg/min versus −0.6 ± 0.5 ml/kg/min; p=0.02).40 However, the improvement in peak VO2 was within the margin of measurement error and was not accompanied by a corresponding improvement in the 6MWT distance or quality of life, so this finding needs to be investigated further. Peak VO2 improvement was not supported by the pharmacokinetic/pharmacodynamic modelling, indicating no relationship between exposure to albiglutide and peak VO2.40 Therefore, current data suggest that GLP-1 RAs do not improve exercise capacity in HFrEF.44
More promising evidence is available regarding GLP-1 RAs in obesity-related HFpEF. The benefits of semaglutide in obese HFpEF patients were established in the landmark STEP HFpEF trial.45 In that trial, patients receiving semaglutide experienced greater reductions in weight (estimated difference −10.7%; p<0.001) and increases in 6MWT distance (estimated difference +20.3 m; p<0.001) compared with those in the control group.45,46 This result was confirmed by the recent pooled analysis of the STEP HFpEF and STEP HFpEF DM trials, which included 1,145 patients. In that analysis, patients in the semaglutide group showed improvement from baseline to week 52 in both body weight (mean 8.4% reduction; p<0.0001 versus placebo) and 6MWT distance (mean 17.1 m; p<0.001 versus placebo).47 Because of the high prevalence of frailty and sarcopenic obesity in the HFpEF population, future studies are required to estimate the proportion of lean body mass loss versus fat loss on GLP-1 RA therapy to identify predictors of the disproportionate loss of muscle mass.45
Conversely, the relative increase in heart rates secondary to GLP-1 RAs may be potentially beneficial in HFpEF due to the high prevalence of chronotropic incompetence in these patients. Another promising strategy for HFpEF patients may be combining exercise training with GLP-1 RAs, mitigating the risk of sarcopenia and frailty and providing a synergistic effect on physical tolerance and quality of life.46 Exercise training can also potentially attenuate gastrointestinal side effects related to GLP-1 RAs.45 Similarly, previous studies showed that caloric restriction with or without aerobic training improved peak VO2 in obese elderly HFpEF patients.48 Future large studies combining comprehensive cardiac rehabilitation programs and intentional weight loss through GLP-1 RAs, caloric restriction and exercise in obesity-related HFpEF will be highly appreciated. It is hoped that ongoing trials like SUMMIT, which is investigating tirzepatide in participants with HFpEF and obesity, will provide more evidence on incretin-based medications in HFpEF.49
Effects of GLP-1 RAs on Quality of Life
In addition to reducing the risk of hospital admission and mortality, improving health-related quality of life (HRQoL) is another treatment target in HF. HRQoL is a broad concept that covers individuals’ views on how their disease and treatment affect their overall wellbeing and physical, psychological and social abilities compared to personal expectations.50 Reduced HRQoL in HF is multifactorial and related to frequent readmissions and symptom burden, exercise intolerance, emotional distress, loss of independence and social limitations. In addition to HF-associated factors, HRQoL is also attributable to comorbidities, the major ones being T2D and obesity. Patients with HFrEF and concomitant T2D show consistently lower Kansas City Cardiomyopathy Questionnaire (KCCQ) scores than those without T2D.49 Similarly, higher BMI has detrimental effects on perceived health in HFrEF, which is also highly significant in HFpEF.51–53 Notably, of the different HF phenotypes, HFpEF is associated with the worst HRQoL, whereas among HFpEF patients, the worst HRQoL had been shown in a subgroup with the highest BMI, a higher proportion of at least Grade 2 obesity and T2D.53 Moreover, there was a linear relationship between HRQoL and BMI.53
Better T2D control and weight reduction with GLP-1 RA therapy may be beneficial in terms of patient-reported health status among the HF cohort. However, in specifically designed placebo-controlled studies of GLP-1 RAs among patients with HFrEF, there were no improvements in HRQoL.39,40,43 There may be several possible explanations for the failure of GLP-1 RAs in HFrEF studies. A well-known collider bias, termed the ‘obesity paradox’, should be considered.54 BMI as a metric of obesity has some limitations, such as not being able to differentiate between lean and fat mass, estimate the magnitude of visceral obesity and predict outcomes.55 Likewise, data on mean BMI among HFrEF participants may not reflect the presence and severity of obesity as a target for the potential effectiveness of GLP-1 RAs.
The FIGHT study showed that more vulnerable HF patients with severely reduced HRQoL and perhaps more advanced stages of the disease may require complex care planning to improve their health status rather than solely increasing glucose and fatty acid metabolism to enhance cardiac metabolism.40 Indeed, patients at later stages of HF may respond differently to therapy that improves outcomes in less severe disease states.56 A short duration of treatment may also have prevented significant changes in HRQoL in HFrEF studies.39,43
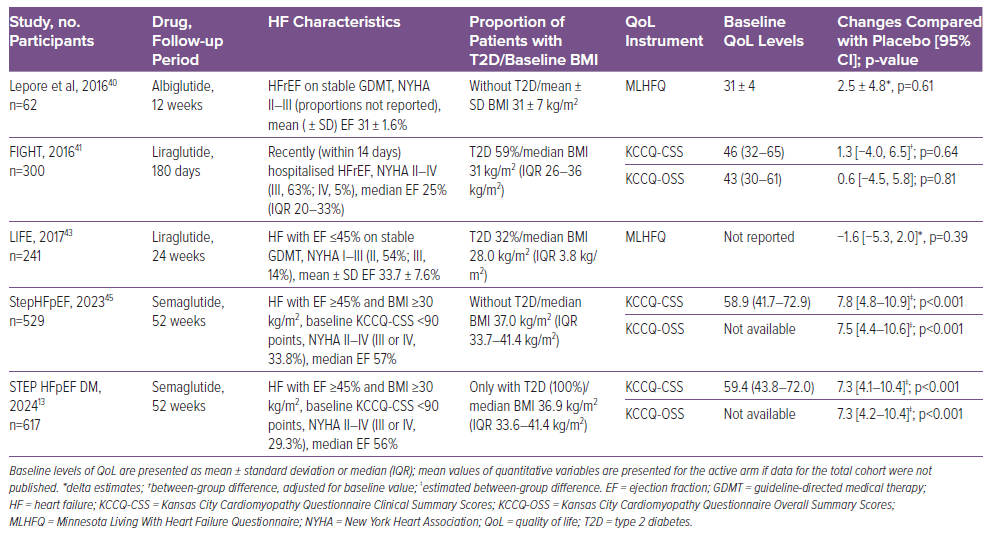
In contrast, among HFpEF cohorts, semaglutide showed clinically meaningful benefits in non-diabetic and diabetic cohorts (Table 1).13,45 In STEP HFpEF, semaglutide improved in KCCQ clinical summary scores (CSS), one of the study’s primary endpoints, by 7.8 points compared with placebo.45 In addition, the KCCQ overall summary score improved by 7.5 points compared with placebo.45 A similar benefit was shown in analyses by baseline KCCQ-CSS tertiles, BMI, and LVEF subgroups.52,57,58 Moreover, the effect was consistent across all HRQoL domains (e.g. physical limitations score, quality of life score, symptom burden score, symptom frequency score and social limitations score), with estimated treatment differences ranging from 6.7 to 9.6 points.52 Similarly, the odds of at least 5-, 10-, 15- and 20-point improvements in all KCCQ domains were 1.6- to 2.9-fold higher among semaglutide- than placebo-treated patients.52 The effect of semaglutide on HRQoL in HFpEF, particularly social and physical function as measured by the KCCQ, is mediated at least in part by the effect on weight reduction.57
Mental health is an integral part of the HRQoL construct and requires attention in HF. HF patients frequently experience low mood, depression and cognitive impairment, with worse estimates shown for the HFpEF phenotype.59 Although some effects of GLP-1 RAs, particularly weight loss, are linked to direct effects of GLP-1 RAs in brain areas responsible for appetite, satiety, food behaviour and other central regulatory mechanisms, data regarding the potential impact of therapy on mental health among HF patients are lacking. Some data suggest neuroprotective properties, and there are ongoing randomised trials among patients with neurological and psychiatric disorders (NCT04466345).60–62 In addition, published reports suggest efficacy of GLP-1 RAs against weight gain related to the use of antipsychotic medication that may affect a patient’s feelings.63 Once-weekly GLP-1 RAs may also improve HRQoL through greater treatment satisfaction.64
In contrast with prior anti-obesity medications, concerns about suicide intention have not been confirmed for GLP-1 RA therapy.65,66 Of all adverse events reported, psychiatric side-effects of GLP-1 RAs are not frequent and account for 1.2–4.4% of events.67,68 Still, the presence of GLP-1 RA-specific psychiatric adverse events that may also be related to drug intolerance requires the development of a strategy to overcome this barrier for the better implementation of GLP-1RA therapy.67
Controversy Regarding Effects of GLP-1 RAs in Heart Failure: Direct Drug Effects or Due to Weight Loss?
Previously described cardiovascular effects of GLP-1 RAs are primarily indirect and include enhanced endothelial function and myocardial metabolism, natriuresis and anti-inflammatory and blood pressure-lowering effects. Given that GLP-1 RAs result in weight loss and the pronounced effects of weight loss on haemodynamic disturbance and cardiac reverse remodelling in obese HF patients, the question is, to what extent is the effect of the drugs related to a direct effect on the cardiovascular system in HF?
A study of 5,067 overweight and obese T2D patients failed to demonstrate an association between mild weight reduction and improved cardiac function.69 In obese HFpEF patients, weight-reduction interventions have significant potential for improving HRQoL.70 A systematic review of 22 studies investigated the effect of intentional weight loss in overweight and obese patients with HF and demonstrated that all forms of weight loss (lifestyle changes, pharmacotherapy or bariatric surgery) are likely to result in significant improvements of symptoms and HRQoL in HF patients.70 In the STEP HFpEF trial, higher changes in KCCQ-CSS were linearly associated with weight loss with semaglutide (5.9-point increase in KCCQ-CSS per 10% body weight loss).45 Remarkably, the recent SELECT trial proved the cardiovascular efficacy of subcutaneous semaglutide in reducing the primary cardiovascular composite endpoint (death from cardiovascular causes, non-fatal MI or non-fatal stroke) in both overweight and obese patients with established atherosclerotic CVD.71 These data, together with the finding of no effect of prior weight-reduction strategies on hard outcomes in HF, signify very likely beneficial effects of GLP-1RAs that are independent of weight loss. Moreover, a counterintuitive reduction in N-terminal pro B-type natriuretic peptide concentrations on top of weight reduction has been shown in both STEP HFpEF trials, indicating potential direct cardiac effects of the therapies.72 Still, whether GLP-1RAs in HFpEF exert a disease-modifying effect independent of weight loss remains to be investigated.
The beneficial effects of GLP-1 RAs on HRQoL and other outcomes in HFpEF are likely due to pleiotropic impacts, related or not to weight reduction, such as decongestion (mainly due to the reduction of increased plasma volume associated with obesity and epicardial constrain), reverse cardiac remodelling (mainly due to the decrease in epicardial fat and regression of left atrial and LV myopathy, lowering intracardiac pressure) and anti-inflammatory actions, among others. Additional mechanistic studies are needed to confirm these assumptions.
Future Directions
Recently, Sundaram presented the results of PRAISE HFpEF DM.73 This was a multicentre observational cohort study from 170 hospitals across the US that investigated the effects of GLP-1 RAs on clinical outcomes, a composite of HF hospitalisation and all-cause mortality, in 1,024 obese HFpEF patients compared with 796 controls receiving dipeptidyl peptidase inhibitor/sulphonylurea therapy. The study showed a 20% risk reduction of the primary outcome in the GLP-1 RA arm (HR 0.80; 95% CI [0.68–0.99]). Furthermore, a prespecified subgroup analysis of HF individuals from the SELECT trial evaluating the effects of semaglutide on cardiovascular outcomes in people with overweight or obesity, which included 2,273 HFpEF and 1,347 HFrEF patients, showed that the therapy was associated with a reduction in the HF composite outcome (cardiovascular death or HF hospitalisation/emergency visit) across LVEF, suggesting that GLP-1 RAs may improve outcomes at least in obese HF patients regardless of diabetes status.74 However, RCTs are still needed to investigate the effects of GLP-1 RAs on HF outcomes, mortality and clinical outcomes in both HFpEF and HFrEF.
Currently, there is an ongoing randomised double-masked placebo-controlled trial, the SUMMIT trial, studying the efficacy and safety of tirzepatide, a dual agonist of glucose-dependent insulinotropic polypeptide and GLP-1 receptors, versus placebo in HFpEF patients with obesity (NCT04847557). The study’s co-primary endpoints are changes in HRQoL and the composite of cardiovascular death and HF events. That study will provide insights into the potential of GLP-1 RAs to modify the disease course in HFpEF patients.
Conclusion
The effects of GLP-1 RAs on HF surrogate endpoints vary in HF patients across the LVEF spectrum. In HFrEF, the administration of GLP-1 RAs did not improve LV systolic function. In HFpEF, the effects of the drugs on cardiac function and structure have not yet been investigated. GLP-1 RA therapy significantly improved both exercise capacity and HRQoL in individuals with obesity-related HFpEF regardless of diabetes status, but had no effect on these parameters in in HFrEF patients. Data from observational studies and subgroup analyses of RCTs show that GLP-1 RAs reduce HF outcomes and mortality in obese HF patients across the LVEF spectrum. However, given the neutral effect of GLP-1 RAs on surrogate HF outcomes in HFrEF and a potentially increased risk of arrhythmias and HF-related hospitalisations, conducting further large-scale trials in the HFrEF cohort seems complicated. Still, it may be relevant to perform pilot studies to address whether the therapy will benefit selected HFrEF cohorts with obesity.










